How does sunscreen work?
Sunscreens are composed of UV filters, which disperse UV rays so they are not absorbed by the our skin. These UV filters can be made of chemicals or minerals.
Chemical UV filters use chemicals like Oxybenzone and Octinoxate to deflect UV rays from our skin by converting light energy into heat energy, which is then released by our skin. These chemicals harm not only corals, but also us humans.
Mineral UV filters instead use Zinc Oxide and Titanium Dioxide to scatter the UV rays, thus blocking them from being absorbed by our skin. These ingredients occur naturally and do not harm humans or corals.
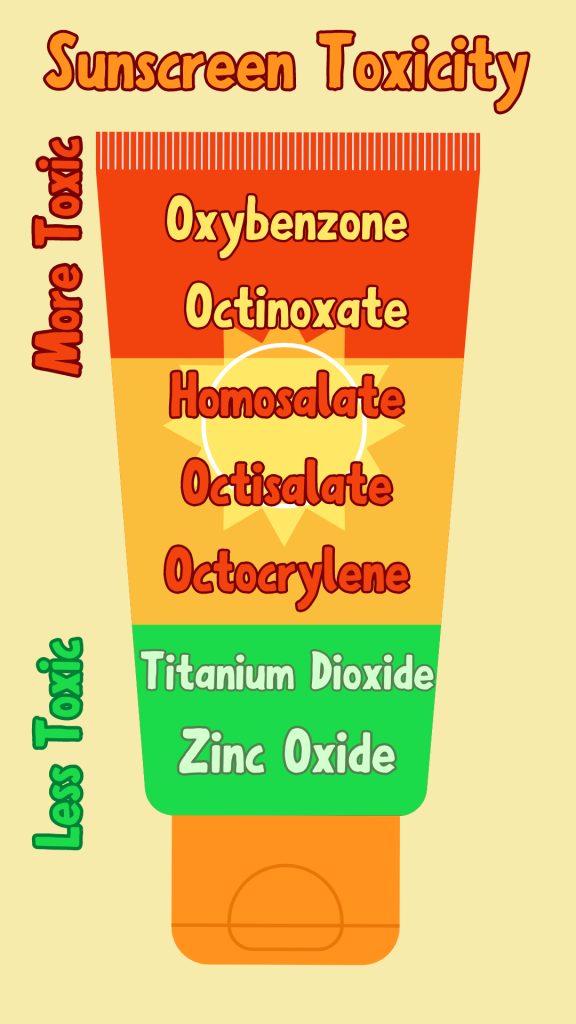
Why are chemical sunscreens so bad?
Well, let’s look at those chemicals they use as UV filters:

Oxybenzone is found in over 3500 sunscreen products worldwide, making it very common in sunscreens. Oxybenzone is found in many aquatic environments and has been shown to disrupt coral reproduction, cause coral bleaching, and damage coral DNA. Oxybenzone is absorbed through the skin and can be found in urine as quickly as 30 minutes after application, allowing it to bioaccumulate in our bodies. Bioaccumulation is the process of a build-up of certain toxins or chemicals in our bodies that we were exposed to over time; the little bit left behind in our system accumulates until there is a large amount. Oxybenzone has been found in bird eggs, fish, coral, humans, and other marine animals. Oxybenzone can be biomagnified if we eat a fish with high concentrations of oxybenzone, giving us a large amount of that toxic chemical in our body. This is biomagnification, where one predator animal ingests a prey organism with a high concentration of some toxin build up in its body, passing the toxins onto the predator (which could be human) and consuming that prey. Essentially, biomagnification means chemicals like Oxybenzone may increase in concentration in the tissues of organisms as it travels up the food chain. The more Oxybenzone washed into the water, the more likely it is consumed by a smaller organism which is then consumed by a human who has ingested large amounts of a toxin. The abundance of Oxybenzone in our waters makes this more likely; it is found in parts per trillion concentrations off the coast of Barrow, Alaska, U.S.A, to parts per billion on coral reefs in the Caribbean, Pacific, and Red Sea. One of the highest concentrations measured in the marine environment was in Trunk Bay in the U.S. Virgin Islands National Park, St. John Island, U.S. Virgin Islands, where the beach can have up to 5,000 people daily.

Octinoxate is absorbed directly through the skin by applying sunscreens and other personal care products. It has been shown to absorb less into the skin, leaving more to wash into the water. Octinoxate can potentially be stored in the body long-term as fat tissue or in lipid-rich tissue due to it being a fat-soluble chemical. Octinoxate is also at risk of being bioaccumulated in organisms and biomagnified through the food web. A number of aquatic and marine species have been discovered to be contaminated, from carp, catfish, eel, white fish, trout, barb, chub, perch, and mussels to coral, mahi-mahi, dolphins, sea turtle eggs, and migratory bird egg. Octinoxate is also found in large concentrations; in coral reef environments, Octinoxate can reach more than 10 parts per billion. Along the west coast of Maui in 2015, in Hawaii, 11 coral reef sites that were sampled had Octinoxate concentrations from 6.9 parts per trillion to 1,516 parts per trillion.
How does sunscreen end up in marine habitats?
- When we shower off from the day, the residual sunscreen on our skin washes into the water and down the drain, eventually flowing into our oceans.

2. Sunscreen is also leeched directly into our environment when we swim in the ocean.

Once Sunscreen is in marine environments, how does it affect marine life?
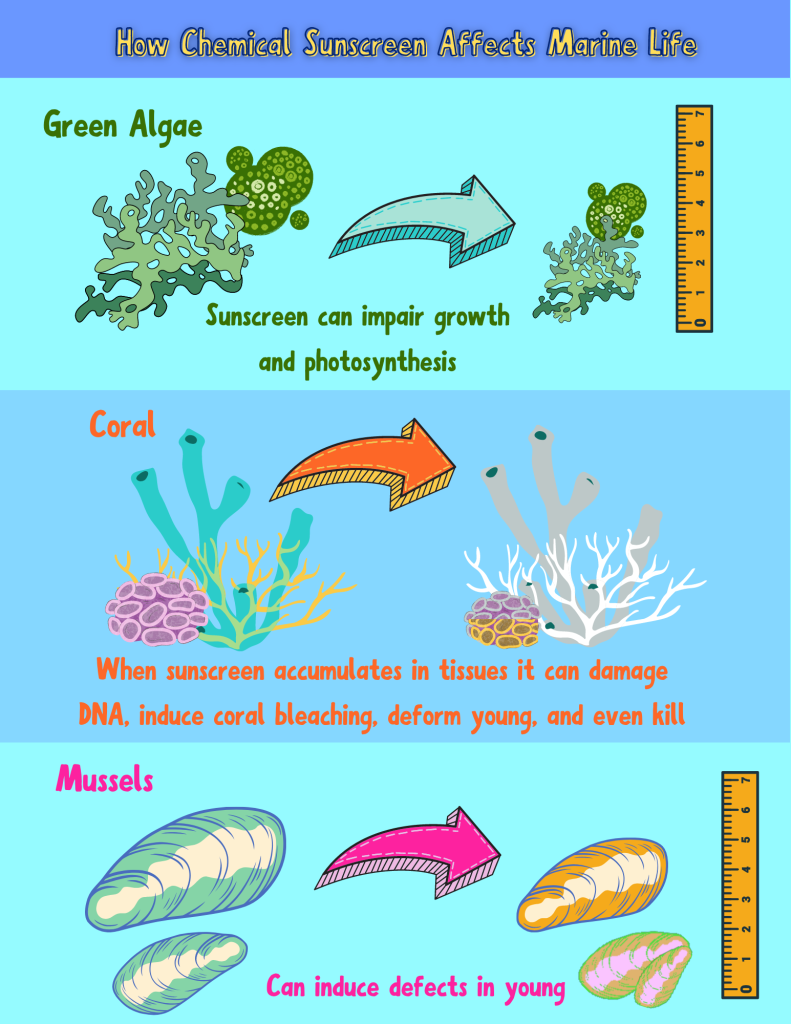

Yikes, so how do I avoid these chemicals that harm not only me, but also corals?
You can avoid the bad side effects of chemical sunscreens by opting to consume mineral sunscreen. Mineral sunscreens use Zinc Oxide and Titanium Dioxide, both naturally occurring minerals, to reflect UV rays and protect the skin. Unlike chemical sunscreen, Zinc Oxide and Titanium Dioxide can reflect both UVA and UVB rays. The graphic to the left demonstrates how UV rays bounce off the skin when they are reflected by mineral sunscreen and how they are absorbed in the absence of sunscreen.
Key Takeaway: Remember, to look for sunscreens with Zinc Oxide and Titanium Dioxide or opt for alternative sun protection, such as brimmed hats, sun shirts, sun leggings, UV sunglasses, using an umbrella, and seeking shade from 10 AM to 2 PM (prime sun-light hours).

Recommendations!
Need some recommendations?
Use code : reefrelief to receive 10% off your Stream2Sea order.
We also recommend sunscreens from Raw Elements, Little Hands Hawaii, All Good, and Raw Love.
Check out Waterlust for long lasting, durable sun gear!